Whiskers are single-crystal materials that grow in a filament-like or needle-like shape. They resemble short fibers, but their dimensions are much smaller. Whisker materials can be classified into organic whiskers and inorganic whiskers. Common inorganic whisker materials include SiC, Si₃N₄, K₂Ti₁₃O₃₀, Al₁₈B₄O₃₃, ZnO, MgO, Al₂O₃, and CaCO₃. The atomic structure inside a whisker is highly ordered. Their diameters are typically at the micrometer scale. As a result, whiskers contain almost no defects commonly found in bulk crystals, such as dislocations, voids, or structural imperfections. Due to the absence of these defects, the strength and elastic modulus of whiskers are close to the theoretical values of perfect crystals.
Therefore, whiskers exhibit high strength, high modulus, corrosion resistance, and high-temperature resistance. They are widely used in paper, rubber, friction materials, and concrete.
Main Physical Parameters of Inorganic Whiskers
| Whisker | Density /(g/cm³) | Diameter /μm | Length /μm | Tensile Strength /GPa | Elastic Modulus /GPa | Mohs Hardness | Thermal Expansion Coefficient /(10⁻⁶/°C) | Melting Point /°C | Heat Resistance /°C |
|---|---|---|---|---|---|---|---|---|---|
| SiC | 3.18 | 0.05–7 | 5–200 | 21 | 490 | 9 | 4.0 | 2690 | 1600 |
| Si₃N₄ | 3.2 | 0.1–1.6 | 5–200 | 14 | 380 | 3.0 | — | 1900 | 1700 |
| K₂Ti₄O₉ | 3.3 | 0.1–1.5 | 10–100 | 7 | 280 | 4 | 6.8 | 1370 | 1200 |
| Al₁₈B₄O₃₃ | 2.93 | 0.5–1 | 10–20 | 8 | 400 | 7 | 4.2 | 1950 | 1200 |
| ZnO | 5.78 | 5 | 2–300 | 10 | 350 | 4 | 4.0 | 1720 | — |
| MgO | 3.6 | 3.0–10 | 200–300 | — | — | — | 13.5 | 2850 | 2800 |
| Al₂O₃ | 3.96 | — | — | 21 | 430 | — | — | 2040 | — |
| CaCO₃ | 2.8 | 1–5 | 20–60 | — | — | 3 | — | 759 | 640 |
| CaSO₄ | 2.69 | 1–4 | 100–200 | 20.5 | 178 | 3–4 | — | 1450 | — |
Calcium Carbonate Whiskers

Calcium carbonate whiskers (CW) are white powders. Under microscopic observation, they appear as needle-like or fibrous single crystals. The typical length is about 20–30 μm, and the diameter is about 0.5–1.0 μm. The melting point is approximately 759 °C, and the density is 2.86 g/cm³.
Calcium carbonate whiskers are a new type of short-fiber inorganic material. They feature high whiteness, excellent filling performance, and high tensile strength. Different morphologies of calcium carbonate whiskers can be obtained by adjusting thermodynamic conditions such as pressure, temperature, and concentration, or by adding crystal growth modifiers to the solution. Based on crystal structure, calcium carbonate whiskers can be divided into three types: calcite-type, aragonite-type, and vaterite-type.
Development Status of Calcium Carbonate Whiskers
Calcium carbonate whiskers are low in cost, which helps reduce overall production expenses. They are commonly used as reinforcing and toughening agents or functional inorganic fillers in cementitious materials, plastics, coatings, and friction materials.
Calcium carbonate whiskers are considered a new type of filler. Japanese companies, such as Maruo, were among the earliest to commercialize calcium carbonate whisker products. Development in China started relatively late. However, over the past decade, both research outcomes and commercial products have increased rapidly.
Preparation Methods of Calcium Carbonate Whiskers
The main preparation methods include carbonation, double decomposition reaction, urea hydrolysis, sol–gel method, and gravity crystallization. Among them, the carbonation method is simple, mature, and energy-efficient. It is currently the mainstream industrial process.
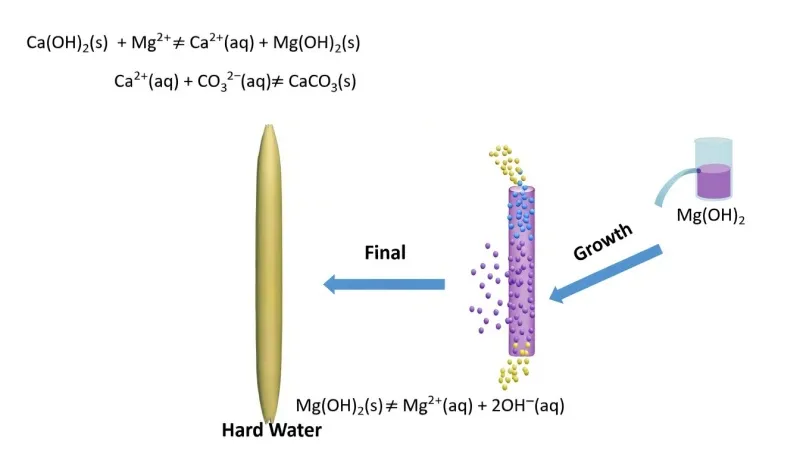
Characteristics of Calcium Carbonate Whisker Preparation Techniques
| Method | Crystal Control Agent | Advantages | Disadvantages |
|---|---|---|---|
| Carbonation method | Required | Simple process, suitable for industrial production | Requires control agents,容易 to introduce impurities |
| Metathesis / Double decomposition method | Not required | High aspect ratio, high purity | Low yield, long reaction cycle |
| Urea hydrolysis method | Not required | High surface smoothness, high purity | Small aspect ratio, high energy consumption |
| Calcium bicarbonate decomposition method | Not required | Longer whiskers, relatively high purity | Poor uniformity, large whisker diameter |
| Sol-gel method | Required | Easy process control, good uniformity | Easily introduces impurities |
| Ultragravity / Hypergravity method | Required | Large aspect ratio, short production cycle | Requires special equipment, high investment |
Carbonation Method
In the carbonation method, carbon dioxide is introduced into a pre-prepared calcium hydroxide suspension. Crystal shape control agents are then added to form calcium carbonate precipitates, typically aragonite-type calcium carbonate. This method is similar to the gas–liquid process used in the industrial production of precipitated calcium carbonate. Therefore, it is also referred to as the gas–liquid method. It is the most widely studied method both domestically and internationally.
In large-scale industrial production, two key issues must be addressed. First, the CO₂ flow rate and its dispersion in the slurry must be carefully controlled. This avoids excessive local supersaturation, which may lead to the formation of calcite or calcium magnesium carbonate impurities. Second, the recovery and reuse of crystal control agents used during carbonation remain a challenge.
Chemical Precipitation Method
The chemical precipitation method involves reacting a carbonate solution of suitable concentration with a soluble or slightly soluble calcium salt solution under controlled conditions to produce calcium carbonate precipitates. In most cases, this method produces calcite-type calcium carbonate. Aragonite-type calcium carbonate can only be synthesized by strictly controlling reaction conditions.
Double Decomposition Method
The double decomposition method uses soluble calcium salts and carbonate salts that react slowly under controlled temperature and stirring conditions, often through dropwise addition. Calcium salts are typically CaCl₂. Carbonate salts commonly include Na₂CO₃ or K₂CO₃. This method is environmentally friendly and simple. The resulting whiskers have a relatively high aspect ratio, smooth surface, and high purity. However, the yield is low and the reaction time is long.
It is important to control the CaCl₂ concentration. If it is too high, local Ca²⁺ supersaturation will occur. This increases the crystallization driving force and promotes the formation of stable calcite rather than aragonite.
Heating Ca(HCO₃)₂ Solution Method
In this method, a calcium bicarbonate solution of a certain concentration is heated to a specific temperature. Ca(HCO₃)₂ decomposes into water, carbon dioxide, and calcium carbonate. Strict control of stirring speed and reaction temperature is required to ensure proper whisker growth.
Continuous Addition of Calcium Hydroxide Method
This method separates nucleation from whisker growth. Under the induction of magnesium chloride, a large number of nuclei are first formed. Calcium hydroxide is then continuously added. By controlling the addition rate of calcium hydroxide, the calcium ion concentration is maintained at a subsaturated level. This allows calcium carbonate whiskers to grow longer, producing large-sized aragonite-type whiskers.
Supergravity Reaction Crystallization Method
This method is similar to supergravity calcium carbonate synthesis. A magnesium chloride solution of a certain concentration is introduced into a calcium hydroxide slurry under a supergravity field generated by a high-speed rotating packed rotor.
Magnesium chloride acts as a crystal form control agent. Using Ca(OH)₂–CO₂ as the reaction system, calcium carbonate whiskers are prepared by adjusting parameters such as rotor speed, liquid flow rate, gas flow rate, carbonation temperature, and magnesium chloride concentration.
Urea Hydrolysis Method
In the urea hydrolysis method, CO₂ generated during urea hydrolysis reacts with soluble calcium salts to form calcium carbonate whiskers.
The key to this method is controlling reaction conditions, such as temperature and pressure. This ensures a slow urea hydrolysis rate and provides a low-supersaturation environment suitable for whisker nucleation and growth.
Carboxylic Acid Decarboxylation Method
In this method, acetic acid reacts with calcium hydroxide to form calcium acetate solutions of different concentrations. These solutions undergo constant-temperature hydrolysis. Highly pure aragonite whiskers can be obtained at concentrations ranging from 0.05 to 0.6 mol·L⁻¹. When heated, organic carboxylic acids release CO₂ through decarboxylation. The release rate of CO₂ can be controlled, creating a homogeneous carbonate precipitation environment.
This method requires relatively simple equipment and reaction conditions. However, care must be taken to prevent the formation of calcite-type CaCO₃.
Surface Modification of Calcium Carbonate Whiskers

Purpose of Modification
The main objectives of calcium carbonate whisker modification are:
- To reduce interparticle cohesion and improve dispersion.
- To enhance surface activity.
- To improve compatibility with other materials.
- To enhance acid resistance.
- To control whisker morphology for different applications.
Modification Processes
Dry Treatment
In dry treatment, calcium carbonate whiskers are processed in a high-speed mixer or kneader. Surface treatment agents are added during mixing to coat the whiskers. This method is simple and suitable for continuous and automated production. However, coating uniformity is limited. It is suitable for various coupling agents and organic surface modifiers.
Wet Treatment
In wet treatment, surface modifiers are directly added to a calcium carbonate suspension. This method provides excellent coating effects and is widely used.
Dry treatment allows direct packaging after processing. Wet treatment offers better surface coverage and is particularly suitable for liquid-phase synthesis of nano calcium carbonate. Common surface modifiers include fatty acids (and their salts) and phosphate esters.
Calcium Carbonate Whisker Modification Equipment

Typical equipment used for calcium carbonate whisker surface modification includes a range of high-shear and high-dispersion mills designed to achieve uniform coating, deagglomeration, and controlled energy input:
- Pin Mill: Widely used for dry surface modification of calcium carbonate whiskers. High rotor speed generates strong impact and shear forces, enabling efficient dispersion of whiskers and uniform attachment of coupling agents or fatty acids. Suitable for continuous production and fine control of coating dosage.
- Turbo Mill: Utilizes high-speed rotating blades to create intense turbulence and shear. Turbo mills are effective for simultaneous grinding, deagglomeration, and surface modification, particularly for applications requiring narrow particle size distribution and good flowability.
- Three-Roller Mill: Mainly applied in paste or high-viscosity systems, such as wet-modified whiskers used in inks, coatings, and adhesives. The strong shear force between rollers ensures excellent dispersion and breaks down soft agglomerates, improving surface coverage and consistency.
- Multi-Rotor Mill: Features multiple rotors operating at different speeds, providing repeated impact, shear, and mixing actions. This design enhances modifier–whisker contact efficiency and is suitable for high-throughput industrial modification with stable product quality.
- Auxiliary Equipment: Depending on the process route, surface modification lines may also include high-speed mixers, wet stirred reactors, continuous coating machines, and downstream dryers such as flash dryers or spray dryers. These units help remove residual moisture while minimizing whisker agglomeration.
The selection of modification equipment depends on the modification method (dry or wet), target application, required coating uniformity, and production capacity.
Applications of Calcium Carbonate Whiskers
Plastics

Calcium carbonate whiskers are commonly used as fillers in PVC, PS, PP, ABS, and other engineering plastics. They improve thermal stability, impact resistance, stiffness, and mechanical strength. In addition, the rheological properties of plastics are improved after adding an appropriate amount of whiskers. This is particularly important for high-end plastic applications.
Coatings
Strong steric hindrance exists between carbon and oxygen atoms in calcium carbonate. When added to coatings, this steric effect increases significantly and suppresses sedimentation. The high whiteness of calcium carbonate whiskers enhances coating brightness and gloss without reducing hiding power. Storage stability is significantly improved.
Due to their steric effects, whiskers can shift absorption peaks toward shorter wavelengths. In latex products, they help shield radiation and improve aging resistance. As inorganic fillers, calcium carbonate whiskers improve flexibility, surface smoothness, self-leveling properties, film formation, and permeability of coatings.
Adhesives and Sealants
Calcium carbonate whiskers are widely used as fillers and reinforcing agents in sealants. On one hand, they can partially replace expensive additives, significantly reducing overall production costs. On the other hand, their unique molecular and crystal structure improves the processing performance of silicone sealants. Noticeable enhancements are observed in mechanical properties such as ultimate tensile strength and fracture strength. In industrial production, by controlling the crystal form, morphology, particle size, and surface modification of calcium carbonate whiskers, sealant systems with excellent thixotropic behavior and anti-sagging performance can be obtained.
Inks

The outstanding physicochemical properties of calcium carbonate whiskers make them highly attractive additives in the ink industry. These advantages are mainly reflected in good powder dispersion, high transparency, proper drying behavior, excellent gloss, and strong ink absorption capacity.
Due to their molecular characteristics, calcium carbonate whiskers are commonly used as fillers in resin-based inks. Compared with gelatinous calcium carbonate, whiskers exhibit more stable performance, better color fidelity, broader application adaptability, and the ability to maintain original drying characteristics. As a result, they are often used in industry to replace more expensive gelatinous calcium carbonate.
Papermaking
Calcium carbonate whiskers feature a unique needle-like structure. This structure improves paper filling rate, tensile strength, and folding endurance.
In addition, the high aspect ratio and needle-shaped morphology allow calcium carbonate whiskers to intertwine with paper fibers. This forms structures similar to mullite needles in cement. As a result, the negative impact on fiber-to-fiber bonding is minimal, while paper retention and durability are effectively enhanced.
Friction Materials
Calcium carbonate whiskers offer excellent heat resistance, high strength, high modulus, and low raw material cost. These characteristics significantly improve the wear resistance of friction materials while keeping manufacturing costs lower than those of traditional wear-resistant materials. Studies have shown that calcium carbonate whiskers can prevent the decline of wear resistance and friction coefficient at elevated temperatures. This finding has been widely applied in automotive brake pads and clutch materials.
When calcium carbonate whiskers are incorporated into polyether ether ketone (PEEK), both the friction coefficient and specific wear rate decrease. At the same time, thermal stability, stiffness, and load-bearing capacity are improved. Thermal softening and adhesion during friction are also effectively suppressed. When the whisker content reaches 15 wt%, the specific wear rate is minimized and improves by approximately seven times compared with pure PEEK.

Artificial Bone Materials
Calcium carbonate whiskers implanted into bone tissue are gradually resorbed. After approximately 12 weeks, they become difficult to detect in vivo.
Therefore, calcium carbonate whiskers can serve as reinforcing fillers for biodegradable polymer materials.
Calcium carbonate whiskers are mildly alkaline. When the surrounding pH value is between 9 and 9.5, even if they enter the human body, they will decompose under acidic physiological conditions and pose no harm. This indicates strong potential for biomedical applications, although both domestic and international research in this area remains limited.
Cement Additives
As a material with excellent mechanical properties, calcium carbonate whiskers can fill microcracks and pores in hardened cement paste.
This significantly improves both the mechanical performance and the microstructural integrity of cement-based materials.
Conclusion
Calcium carbonate whiskers are a promising new construction and functional material. Although preparation technologies are not yet fully mature and large-scale applications are still expanding, they offer outstanding cost-performance advantages and strong application potential. They exhibit excellent rigidity, dimensional stability, and thermal stability. When incorporated into plastic products, they combine rigidity with the toughness of polymer materials, thereby expanding the application range of existing plastics.
Due to their remarkable reinforcing and toughening effects, low agglomeration tendency, and good dispersibility, calcium carbonate whiskers are widely used in cement fillers, papermaking, and friction materials. Moreover, their complete crystal structure, low defect density, low cost, and simple preparation process make them ideal fillers for enhancing strength, elastic modulus, alkali resistance, and overall mechanical performance of composite materials

“Thanks for reading. I hope my article helps. Please leave a comment down below. You may also contact Zelda online customer representative for any further inquiries.”
— Posted by Emily Chen